Multifocal Ewing sarcoma. Presentation of a pediatric case.
DOI:
https://doi.org/10.35954/SM2023.42.1.3.e501Keywords:
Bone neoplasms, Ewing sarcoma, Neuroectodermal Tumors, Primitive, Peripheral.Abstract
Introduction: Ewing's sarcoma is a malignant neoplasm of mesenchymal origin. At the time of diagnosis 75% of the cases are localized.
Objective: to report a case that, due to its multifocal presentation, generated diagnostic difficulties.
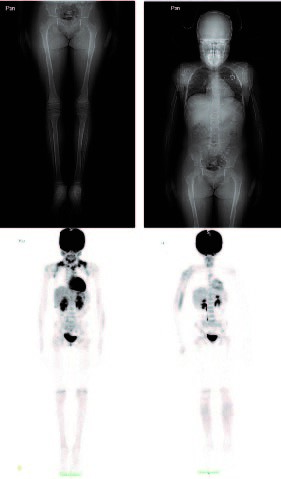
Clinical case: 6-year-old girl. She consulted for right hand trauma after a fall from her height 24 hours earlier, with edema, warmth and erythema in the right hand and fist, with preserved mobility. No fever. X-ray: increase in the diameter of the 3rd metacarpal, frosted image, no traces of fractures. Admitted with cellulitis. Mild microcytic anemia, hypochromic. C-reactive protein 82mg/l. Receives intravenous clindamycin 72 hours, completes 14 days orally. Persistence of alterations in fist and right hand, adds tumor of nasal root with deviation of the axis, painless. Alkaline phosphatase, lactate dehydrogenase, phosphatemia, normal calcemia. MRI: morphostructural alteration of radius, olecranon and 3rd metacarpal, fracture of olecranon and radius, periosteal reaction. Pet-Scan: extensive bone lesion in facial mass, tibiae, ulnae, right humerus and clavicle. Biopsy 3rd metacarpal: small cell tumor, blue round, CD 99 and vimentin positive. Polychemotherapy and radiotherapy were started without complications.
Conclusions: it is frequent that the initial clinical manifestations are confused with more frequent entities, such as post-traumatic and/or inflammatory, as occurred in this case. Subsequently, the appearance of new lesions and compromise of the general condition guided the diagnostic approach of the tumor pathology. Confirmation requires anatomopathological study with immunohistochemical study. The presence of bone metastases constitutes a poor prognostic factor and hinders the therapeutic approach.
Received for review: January 2023.
Accepted for publication: March 2023.
Correspondence: Bulevar Artigas 1590. Montevideo, Uruguay. Tel: (+598) 27091443.
Contact e-mail: anitacasuriaga7@gmail.com
Downloads
Metrics
References
(1) Sanchez-Saba JE, Abrego MO, Albergo JI, Farfalli GL, Aponte-Tinao LA, Ayerza MA, et al. Sarcoma de Ewing óseo: Enfoque multidisciplinario y resultados oncológicos en 88 pacientes. Medicina (B. Aires) [Internet]. 2020 Feb [citado 2023 Ene 22]; 80(1): 23-30. Disponible en: http://www.scielo.org.ar/scielo.php?script=sci_arttext&pid=S0025-76802020000100004&lng=es
(2) Dragovetzky A, Bagnara E, Nastrid M. Localización infrecuente de un sarcoma de Ewing en pediatría. Archivos Argentina Pediatría 2019; 117(3):206-207. Disponible en: https://www.sap.org.ar/docs/publicaciones/archivosarg/2019/v117n3a32.pdf DOI: https://doi.org/10.5546/aap.2019.206
(3) Whelan J, McTiernan A, Cooper N, Wong Y, Francis M, Vernon S, et al. Incidence and survival of malignant bone sarcomas in England 1979-2007. Int J Cancer 2012; 131(1):17-50. https://doi.org/10.1002/ijc.26426 DOI: https://doi.org/10.1002/ijc.26426
(4) Mosquera Betancourt G, Hernández González EH, Hernández Cabezas I, Quintero Martínez O. Sarcoma de Ewing extraesquelético del raquis dorsal: presentación de dos casos. Rev Cubana Neurol Neurocir [Internet]. 2014 Feb [citado 2023 Ene 22]; 4(2):166-73. Disponible en: file:///C:/Users/usuario/Desktop/Downloads/Dialnet-SarcomaDeEwingExtraesqueleticoDelRaquisDorsal-4790488.pdf
(5) Randall L, Calvert G, Spraker H, Lessnick S. Familia de tumores del sarcoma de Ewing (ESFT). [sitio Web]. Utah: Liddy Shriver Sarcoma Initiative; 2011 [citado Ene 22]. Disponible en: http://sarcomahelp.org/translate/es-sarcoma-ewing.html
(6) Villalta J. Sarcoma de Ewing. Revista médica de Costa Rica y Centroamérica 2015; LXXII(617):695-704.
(7) Robert K. Tumores óseos malignos. En: Terry Canale S, editor. Campbell´s Operative Orthopaedics. 10ma ed. Philadelphia: Mosby; 2003. p. 836-38.
(8) Marquina G. Sarcomas óseos. [sitio Web]. SEOM Sociedad Española de Oncología Médica. 2023. [citado marzo 2023]. Disponible en: https://www.seom.org/info-sobre-el-cancer/oseosewing?showall=1&showall=1
(9) Rodriguez-Galindo C, Navid F, Khoury J, Krasin M. Ewing Sarcoma Family of Tumors. In: Pappo A (eds). Pediatric Bone and soft Tissue Sarcomas. Pediatric Oncology. Springer-Verlag : Berlin Heidelberg, 2006. Capter 9. p.181-217. https://doi.org/10.1007/3-540-29447-3_9 DOI: https://doi.org/10.1007/3-540-29447-3_9
(10) Ali SA, Muhammad AT, Soomro AG, Siddiqui AJ. Extra osseous primary Ewing's sarcoma. J Ayub Med Coll Abbottabad 2010; 22(3):228-9. PMID: 22338463.
(11) Hoffman H, Suar P. Sarcoma de Ewing en la Infancia. [sitio Web]. Sociedad Iberoamericana de información científica. 2000. [citado enero 2022]. Disponible en: https://www.siicsalud.com/des/expertoimpreso.php/20583
(12) Ginsberg JP, Woo SY, Hicks MJ, Horowitz ME. Ewing’s sarcoma family of tumors: Ewing’s sarcoma of bone and soft tissue and the peripheral primitive neuroectodermal tumors. In: Principles and Practice of Pediatric Oncology. 4th ed., Philadelphia, 2002.
(13) Rey G. Sarcomas en Pediatría. Rev Hosp Niños BAires 2011; 53(241):56-69. Disponible en: http://revistapediatria.com.ar/wp-content/uploads/2012/03/con375-56-69.Rey_.pdf [Consulta 23/01/2023].
(14) Granowetter L, Womer R, Devidas M, Krailo M, Wang C, Bernstein M, et al. Dose-intensified compared with standard chemotherapy for non-metastatic Ewing sarcoma family of tumors: a Children’s Oncology Group Study. J Clin Oncol 2009; 27(15):2536-41. doi: 10.1200/JCO.2008.19.1478. DOI: https://doi.org/10.1200/JCO.2008.19.1478
(15) Parasuraman S, Langston J, Rao BN, Poquette CA, Jenkins JJ, Merchant T, et al. Brain metastases in pediatric Ewing sarcoma and rhabdomyosarcoma: the St. Jude Children’s Research Hospital experience. J Pediatr Hematol Oncol 1999; 21(5):370-7. doi: 10.1097/00043426-199909000-00007. DOI: https://doi.org/10.1097/00043426-199909000-00007
(16) Gregianin L, Scopinaro M, Rose A, Boldrini E, Ferman S, Petrilli AS, et al. A multicentric study of interval compressed multiagent chemotherapy and metronomicchemotherapy for patients with Ewing sarcoma family of tumors: The Latin American Pediatric Oncology Group trial. J Clin Oncol 2015; 33(15)suppl I: TPS10079. DOI: https://doi.org/10.1200/jco.2015.33.15_suppl.tps10079
(17) Chaber R, Arthur CJ, Depciuch J, Tach K, Raciborska A, Michalak E, et al. Distinguishing Ewing sarcoma and osteomyelitis using FTIR spectroscopy. Sci Rep. 2018; 8:15081. doi:10.1038/s41598-018-33470-3. DOI: https://doi.org/10.1038/s41598-018-33470-3
(18) Mankin HJ. Ewing sarcoma. Curr Opinion Ortho. 2000; 11:479. DOI: https://doi.org/10.1097/00001433-200012000-00010
(19) Mendenhall CM, Marcus RB Jr, Enneking WF, Springfield DS, Thar TL, Million RR. The prognostic significance of soft tissue extension in Ewing’s sarcoma. Cancer 1983; 51(5):913-7. doi: 10.1002/1097-0142(19830301)51:5<913::aid-cncr2820510525>3.0.co;2-0. DOI: https://doi.org/10.1002/1097-0142(19830301)51:5<913::AID-CNCR2820510525>3.0.CO;2-0
(20) Martínez V. Anatomía patológica de Sarcoma de Ewing. Hospital San Juan de Dios, Santiago, Chile. Medwave. 2001; 1(03). doi:10.5867/MEDWAVE.2001.03.3476. DOI: https://doi.org/10.5867/medwave.2001.03.3476
(21) Bannasch H, Eisenhardt S, Grosu A, Heinz J, Momeni A, Stark B. The diagnosis and treatment of soft tissue sarcomas of the limbs. Dtsch Arzte blInt 2011; 108(3):32-38. doi: 10.3238/arztebl.2011.0032. DOI: https://doi.org/10.3238/arztebl.2011.0032
(22) Hernández EH, Mosquera Betancourt G, Quintero Martínez O, Hernández Cabezas I. Sarcoma de Ewing. Archivo Médico de Camagüey [Internet] 2013; [citado 2023 Ene 22]; 17(5):623-640. Disponible en: https://www.redalyc.org/pdf/2111/211129323012.pdf
(23) Albano D, Dondi F, Schumacher RF, D'Ippolito C, Porta F, Giubbini R, et al. Clinical and Prognostic Role of 18F-FDG PET/CT in Pediatric Ewing Sarcoma. J Pediatr Hematol Oncol. 2020 Mar; 42(2):e79-e86. doi: 10.1097/MPH.0000000000001518. Balmant N, Silva N, Santos M, Reis R, Camargo B. Atrasos en el sistema de salud de niños, adolescentes y adultos jóvenes con tumores óseos, en Brasil. J Pediatr (Rio J.). 2019; (6):744-751. DOI: https://doi.org/10.1097/MPH.0000000000001518
(24) Haeusler J, Ranft A, Boelling T, Gosheger G, Braun-Munzinger G, Vieth V, et al. The value of local treatment in patients with primary, disseminated, multifocal Ewing sarcoma (PDMES). Cancer 2010 Jan 15; 116(2):443-50. doi: 10.1002/cncr.24740. DOI: https://doi.org/10.1002/cncr.24740
(25) Ladenstein R, Pötschger U, Le Deley MC, Whelan J, Paulussen M, Oberlin O, et al. Primary disseminated multifocal Ewing sarcoma: Results of the Euro-EWING 99 trial. J Clin Oncol 2010 Jul 10; 28(20):3284-91. doi: 10.1200/JCO.2009.22.9864. DOI: https://doi.org/10.1200/JCO.2009.22.9864
(26) Zöllner SK, Amatruda JF, Bauer S, Collaud S, de Álava E, DuBois SG, et al. Ewing Sarcoma Diagnosis, treatment, clinical challenges and future perspective. J Clin Med. 2021 Apr 14; 10(8):1685. doi: 10.3390/jcm10081685. DOI: https://doi.org/10.3390/jcm10081685
Downloads
Published
Versions
- 2023-05-30 (2)
- 2023-04-26 (1)
How to Cite
Issue
Section
License
Copyright (c) 2023 Evelyn Villalba , Ana Casuriaga, Agustín Dabezies , Fabiana Morosini y Gustavo Giachetto. The author retains his copyright and assigns to the journal the right of first publication of his work, which will be simultaneously subject to the Creative Commons Attribution 4.0 International License that allows sharing the work as long as the initial publication in this journal is indicated.

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
Until 2024 we use the Creative Commons Attribution/NonCommercial Attribution 4.0 International License https://creativecommons.org/licenses/by-nc/4.0/deed.es. Which states that: you are free to share, copy and redistribute the material in any medium or format, as well as to adapt, remix, transform and build upon the material. Under the following terms:
Attribution: you must give proper credit , provide a link to the license, and indicate if changes have been made . You may do so in any reasonable manner, but not in such a way as to suggest that you or your use is endorsed by the licensor.
NonCommercial: you may not use the material for commercial purposes.
As of 2025 authors retain their copyright and assign to the journal the right of first publication of their work, which shall simultaneously be subject to the license https://creativecommons.org/licenses/by-nc-sa/4.0/deed.es that permits sharing, copying and redistribution of the material in any medium or format provided that initial publication in this journal is indicated. Adapt, remix, transform and build upon the material. If you remix, transform, or build from the material, you must distribute your contribution under the same license as the original and may not make use of the material for commercial purposes.
Under the following terms:
1. Attribution: you must give proper credit, provide a link to the license, and indicate whether changes have been made. You may do so in any reasonable manner, but not in such a way as to suggest that you or your use is endorsed by the licensor.
2. NonCommercial: you may not use the material for commercial purposes.
3. ShareAlike: if you remix, transform or build upon the material, you must distribute your contribution under the same license as the original.
PlumX Metrics





















